Patient-Specific Cranial Implants
SU-POR® Patient-Specific Implants are individualized to match each patient’s unique anatomy and surgical needs. Designed in collaboration with the patient’s surgeon, these implants ensure a precise fit, optimal function, and aesthetic restoration.
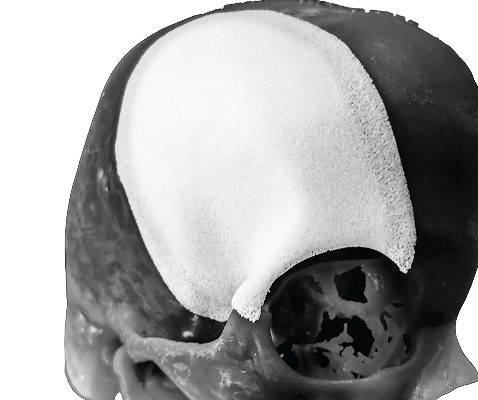
Your Design, Crafted and Delivered with Precision
Expert Planning
Complementary planning session with our experienced engineering team.
Risk Reduction
Sterilized in-house via Ethylene Oxide, reducing hospital liability and resources
Fast Delivery
Is shipped in as little as 8 business days, following Design Approval and Purchase Order
Complete Kit
Sterile implant(s), non-sterile medical model, and non-sterile sample for patient consultation
Start Your Order Today
Our Key Features
Precise Fit
Each device is individually crafted to your patient’s specific needs.
Proven Material
Porous polyethylene, a biocompatible material with over 30 years of proven clinical performance.

Fast Turnaround
Order shipment in as little 8 business days following design approval.
Modification
Malleable, with Retained Shape Memory™ technology. SU-POR® implants allow for easy intraoperative modification with a scalpel blade, scissors, or burring instrument.
Variable Thickness
Variable thickness to match native bone. This ensures the implant matches the patient for premium results.
Virtual Approval Process
Detailed electronic document allows for approval prior to manufacturing, giving our engineers the tools they need to provide the highest quality results.
EO Sterile, Non-pyrogenic
Implant kit contains sterile, non-pyrogenic implant(s), limiting processing risk for the hospital.
MR Safe
This designation by the US FDA provides assurance of no image artifact or distortion of diagnostic MRI imaging.
Suture & Drainage
Sutures may be passed through any part of the implant. Open pores may allow drainage.
Tissue In-Growth
The interconnecting, omnidirectional pore structure allows for fibrovascular and tissue integration, giving the implant its own blood supply.
Application
To meet the need of individual patients in applications of craniofacial reconstructive/cosmetic surgery and repair of craniofacial trauma.
Direct Fixation
Screws may be inserted directly through the implant margins into the bone and may eliminate the need for plate fixation.